The fluorescence of a diamond is the visible glow that occurs when it is exposed to ultraviolet (UV) light. Fluorescence is a naturally occurring phenomenon created from the mineral properties boron and nitrogen. It can appear in many minerals and gems.
Diamond is crystallized carbon. This process occurs about 80-120 miles below the surface of the earth, usually where volcanic or ancient volcanic sites exist. A diamond will become fluorescent if there are traces of the mineral boron present in the earth during the crystallization process.
Nearly all diamonds have some degree of fluorescence, although certain lighting conditions makes this difficult to see with the unaided eye. An ultraviolet (UV) lamp, however, can be used to reveal a soft-colored, fluorescent glow. Diamond fluorescence occurs in varying intensities and colors. The GIA rates the fluorescence of each diamond on a scale ranging from "None" to "Very Strong". Fluorescence is categorized as 'None' when there is no visible reaction under ultraviolet light. It will be categorized as 'Faint', 'Medium', 'Strong', or 'Very Strong' depending on how much glow is observed. According to the GIA Gem Trade Laboratory, strength of fluorescence does not directly correlate to either color or clarity . In other words, it is possible for two diamonds with completely different clarity and color grades to exhibit exactly the same strength of fluorescence.
Swipe left to see more.

The images show the varying levels of fluorescence on diamonds viewed table-down (top image) and table-up (bottom image).
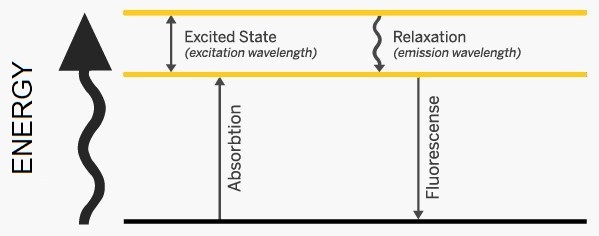
The image shows the relationship between absorption and luminescence (the emission of light by a substance that has absorbed UV radiation.) The figure shows an electron moving from the ground state to an excited state (north arrow) when UV radiation is absorbed at a luminescence center. When the electron moves to a slightly lower state it loses some of its energy (wavy arrow) and no light is emitted. Subsequently, it returns to the groundstate (straight down arrow), releasing energy, or light, as fluorescence.
Diamond fluorescence is graded on a scale established by GIA. Of all the diamonds submitted to GIA for grading, approximately 25% to 35% show some fluorescence. Less than 1% of fluorescent diamond may affect the appearance due to medium, strong or very strong fluorescence. Fluorescence is predominantly blue in glow. At times, you may find diamonds with shades of green, white, pink, yellow, red or orange fluorescence. This is very rare and can have essentially the same impact on the diamond as a blue fluorescent diamond. See the scale table below for more details:
| Fluorescence | Fluorescence Description |
|---|---|
| None, Nil, Negligible | No fluorescence, no influence on color |
| Faint, Slight | Weak fluorescence, not a significant influence on color (barely perceptible) |
| Medium | Average fluorescence, small influence |
| Strong, Very Strong | Strong influence, substantial color influence |
Diamond fluorescence chart.
No soft, blue glow is emitted when the diamond is exposed to UV light. The diamond has no trace elements that cause fluorescence. None fluorescence has the largest premium and is highly desired and expensive.
A slight soft, blow glow is noticed when the diamond is exposed to UV light. The diamond has minor trace elements that cause fluorescence. There is no impact on the diamond. None and Faint fluorescence have an almost identical effect on a diamond's value.
A light blue glow is emitted by a diamond, when exposed to UV light. Medium fluorescence offers an excellent value as it reduces diamond price. It can improve the color of lower grades such as J, K or L.
A deep, blue glow is emitted by the diamond when exposed to UV light. Diamonds with Strong fluorescence can slightly improve lower colors such as J, K or L. Conversely, it can have a negative effect on high color diamonds such as D, E, which can look slightly grayish or hazy.
A very bright blue glow is present when the diamond is exposed to UV light. The diamond's appearance may be effected. Very Strong fluorescence can make a diamond look gray or hazy.
Diamonds that are said to be fluorescent contain particles that emit a visible (usually blue) glow when exposed to the concentrated radiation of a UV lamp or UV lighting. In rare cases, high levels of fluorescence can make stones appear milky or hazy, although for the most part fluorescence does not generally impact beauty or sparkle and can even make some lower colors (I, J, K, L, etc.) appear more colorless or white. Nonetheless, fluorescent diamonds are usually cheaper and provided the effect is not readily visible may offer good value for money.
The subject of diamond fluorescence has been hotly debated in recent years, with trade opinion divided regarding its effect on the appearance and value of diamonds.
As mentioned, submicroscopic structures in the diamonds cause them to emit a visible light, a fluorescence, which is commonly blue in color. Despite the fact that fluorescence is used as an identifying characteristic rather than a grading factor in most laboratory quality reports, its presence in such reports is being used with increasing frequency to determine the pricing of a diamond. In most cases, prices are lowered when a colorless or near colorless diamond fluoresces under UV light, due to a common perception that fluorescence has a negative effect on the appearance of diamonds. However, there are many who believe that fluorescence has no adverse effect; some even contend that it enhances color appearance.
The negative image of fluorescing diamonds can be attributed to the following theories and factors:
Non-fluorescent diamonds are regarded as 'purer'.
Strongly fluorescent diamonds can emit an opaque 'cloud' which affects the clarity and brilliance of the diamond.
GIA lab grading lights emit a small amount of UV radiation, therefore fluorescent diamonds might be assigned a better color grade than they deserve.
Those who refute the notion that fluorescent diamonds are less desirable argue that:
UV light is not present in many viewing environments, so it makes sense to grade color in realistic lighting.
The haziness found in highly fluorescent diamonds is extremely rare.
In some cases, fluorescence can lift color to the naked eye, enhancing the beauty of the diamond.
In Europe, before the age of diamond certification, the most highly regarded diamond color was 'blue white'. This term describes near-colorless to light yellow diamonds with a strong blue fluorescence, stones that were actively sought by merchants thanks to their appealing 'ice' effect.
It was later observed that strong blue fluorescence was a quality sometimes found in hazy stones, a fact that that led dealers in the 1970s to offer what they termed 'milky Ds' (diamonds with a color grade of D, very strong blue fluorescence, low transparency) at significantly reduced prices. This had a knock-on effect during the years that followed: eventually even F grade diamonds and those with a weaker fluorescence were being marked down in price.
Despite a long history of debate on the issue of fluorescence and pricing, there had been no studies that examined the influence of blue fluorescence on the appearance of a diamond under normal viewing conditions until the GIA undertook their 1997 experiment.
Color assessment in grading laboratories takes place under carefully controlled lighting and viewing conditions and mainly with the diamond positioned table-down. However, in a jewellery store or showroom a diamond will normally be examined table-up and in a variety of lighting conditions, as it almost certainly would be when the item is worn. With this in mind, the GIA set up their experiment to study the influence of fluorescence in a range of lighting conditions and by observers from both within and outside the diamond industry.
In the experiment, a range of diamonds with very similar proportions, clarity and symmetry , but differing ranges of blue fluorescence, were assembled. Diamonds of similar color grades were grouped into sets to represent important commercial breakpoints. These sets were then examined in a wide variety of viewing environments, from the controlled conditions found in laboratories to rooms with only natural sunlight. A total of 46 observers were assembled to represent both the diamond industry and the diamond-buying public.
For the non-industry observers, considered to represent the diamond consumer, fluorescence had no effect on color and appearance. For the experienced industry observers, the GIA found that the strength of fluorescence had no widely perceptible effect on the color appearance of diamonds when viewed table-down. What's more, when the diamonds were viewed table-up, as they would be in a jewellery store or showroom, the experienced observers actually reported strongly and very fluorescent diamonds as having a better color appearance than the less fluorescent stones.
The GIA discovered that while the effect of fluorescence on color was most noticeable in stones within lower color grades (I and K), in the marketplace the influence on price is greater in stones with higher color grades (D through H). This challenges the trade perception that fluorescence usually has a negative effect on better-color diamonds. They also found that, contrary to popular belief, fluorescent diamonds aren't as common as non-fluorescent diamonds.
The GIA report concluded that "the diamond industry would be better served by considering each individual diamond on its own visual merits."
For a precise summary of the effects of fluorescence on price, use the chart below. The effect on price varies depending on the degree of diamond color, clarity and fluorescence. For example, according to the chart, an E color diamond of VS1 clarity and Strong Fluorescence will tend to sell for 3-5% less than a like diamond with no fluorescence. Alternatively, a J color diamond of any clarity and Medium fluorescence will typically sell for up to 2% more than a like diamond with no fluorescence. While clarity has no bearing on the appearance of color and fluorescence, it does impact price. This is because customers who gravitate towards higher clarity tend to be more adverse to the presence of fluorescence:
Swipe left to see more.
| Color | Clarity | Fluorescence | |||
|---|---|---|---|---|---|
| Very Strong | Strong | Medium | Faint | ||
| D-F | IF - VVS2 | -10 to -15% | -7 to -10% | -3 to -7% | -1% |
| VS1 - VS2 | -6 to -10% | -3 to -5% | -1 to -2% | 0% | |
| SI1 - I3 | 0 to -3% | 0 to -1% | 0% | 0% | |
| G-H | IF - VVS2 | -7 to -10% | -5 to -7% | -1 to -3% | -1% |
| VS1 - VS2 | -3 to -5% | -2 to -3% | 0 to -2% | 0% | |
| SI1 - I3 | 0% | 0% | 0% | 0% | |
| I-M | IF - I3 | 0 to +2% | 0 to +2% | 0 to +2% | 0% |
Diamond fluorescence effects on price.
While the majority of diamonds with fluorescence exhibit a blue tone under ultraviolet light, other colors are possible. Rare yellow, orange, green and red fluorescence has been documented. These hues can increase the desirability of fancy colored diamonds if they complement their primary hue. For example, a fancy yellow diamond with yellow fluorescence will have an intensity that appeals to many diamond customers, potentially raising the value of the stone.
It's important for you to be happy with the diamond you purchase. As shown, fluorescence can greatly enhance the color and appearance of a diamond and shouldn't be looked upon unfavourably; only in exceptionally rare cases will fluorescence cause the diamond to look milky, oily or hazy. It always comes down to a subjective decision, but best practice would be to consult a diamond specialist before purchasing to ensure you make the right choice.